UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
_____________________
FORM 8-K
_____________________
CURRENT REPORT
PURSUANT TO SECTION 13 OR 15(D)
OF THE SECURITIES EXCHANGE ACT OF 1934
Date of Report (Date of earliest event reported): January 12, 2026
_____________________
(Exact name of registrant as specified in its charter)
_____________________
| (State or other jurisdiction of incorporation) | (Commission File Number) | (IRS Employer Identification No.) | ||||||||||||
| (Address of principal executive offices) | (Zip Code) | |||||||
Registrant’s telephone number, including area code: (617 ) 871-2098
N/A
(Former name or former address, if changed since last report)
_____________________
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | ||||||||
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | ||||||||
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | ||||||||
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) | ||||||||
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading symbol(s) | Name of each exchange on which registered | ||||||||||||
The | ||||||||||||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
| Emerging growth company | |||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02. Results of Operations and Financial Condition.
On January 12, 2026, Akebia Therapeutics, Inc. (the “Company”) issued a press release announcing key corporate updates associated with its Vafseo® (vadadustat) commercial business and providing an outlook on upcoming milestones, including for its next anticipated growth driver, the Company’s mid-stage rare kidney disease pipeline. The Company also provided an overview of expected fourth quarter 2025 Vafseo net product revenue. A copy of the Company’s press release containing this information is furnished as Exhibit 99.1 to this Current Report on Form 8-K (“Report”) and is incorporated herein by reference.
Item 7.01. Regulation FD Disclosure.
Spokespersons of the Company plan to present the information in the corporate presentation attached hereto as Exhibit 99.2 (the “Presentation”) at various meetings beginning on January 12, 2025, including investor and analyst meetings that coincide with the 44th Annual J.P. Morgan Healthcare Conference.
A copy of the Presentation is attached as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated herein by reference.
The information in this Current Report on Form 8-K (including Items 2.02 and 7.01, including Exhibits 99.1 and 99.2) shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Exchange Act or the Securities Act, of 1933, as amended except as expressly set forth by specific reference in such a filing.
By providing the information in Item 7.01 of this Current Report on Form 8-K, including Exhibits 99.1 and 99.2 hereto, the Company is not making an admission as to the materiality of any information herein. The information contained in this Current Report on Form 8-K is intended to be considered in the context of more complete information included in the Company’s filings with the SEC and other public announcements that the Company has made and may make from time to time by press release or otherwise. The Company undertakes no duty or obligation to update or revise the information contained in this Current Report on Form 8-K, although it may do so from time to time as its management believes is appropriate. Any such updating may be made through the filing of other reports or documents with the SEC, through press releases or through other public disclosures.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits
Exhibit No. | Description | |||||||
99.1 | ||||||||
| 99.2 | ||||||||
104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | |||||||
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| AKEBIA THERAPEUTICS, INC. | ||||||||
Date: January 12, 2026 | By: | /s/ John P. Butler | ||||||
| Name: John P. Butler | ||||||||
| Title: President and Chief Executive Officer | ||||||||
Exhibit 99.1
Akebia Therapeutics Announces Corporate Updates and 2026 Pipeline Outlook
Positioned to increase depth of Vafseo prescribing entering 2026 with access to approximately 275,000 patients
First patient dosed in Praliciguat Phase 2 clinical trial studying focal segmental glomerulosclerosis (FSGS)
AKB-097 Phase 2 rare kidney disease basket trial scheduled to begin in 2H 2026 with initial data expected in 2027
CAMBRIDGE, Mass.—January 12, 2026—Akebia Therapeutics, Inc. (Nasdaq: AKBA), a biopharmaceutical company with the purpose to better the lives of people impacted by kidney disease, today announced key corporate updates associated with its Vafseo® (vadadustat) commercial business and provided an outlook on upcoming milestones, including for its next anticipated growth driver, Akebia’s mid-stage rare kidney disease pipeline.
“We enter 2026 in a solid financial position and expect increased demand for Vafseo as we believe existing customers will accelerate adoption of the product and new customers will operationalize Vafseo protocols within their organizations,” said John P. Butler, Chief Executive Officer of Akebia. “We continue to generate post-marketing Vafseo clinical data that will support our goal to make Vafseo standard of care to treat anemia due to chronic kidney disease (CKD) in dialysis. In parallel, we are aggressively progressing our pipeline, including our recently announced rare kidney disease programs, where we will leverage our scientific leadership in nephrology with an aim to help patients in need of new therapies. Our revenue-generating products are the engine driving advancement of our mid-stage pipeline, which, along with continued adoption of Vafseo, we believe can drive significant shareholder value this year and beyond.”
Driving to Standard of Care – 2025 Vafseo Achievements
•Secured broad prescribing access for Vafseo encompassing approximately 275,000 patients on dialysis, which is expected to facilitate additional demand in 2026.
•Supported dialysis organizations’ implementation of modified dosing protocols or pilots now underway at the top 5 dialysis organizations with prescribing access.
•Completed enrollment in VOICE, a large Phase IV trial of over 2,100 patients evaluating Vafseo TIW against standard-of-care erythropoietin stimulating agents (ESAs) using a hierarchical composite endpoint of all-cause mortality and all-cause hospitalization.
VOICE topline results are expected in early 2027 with the potential to help establish Vafseo as a standard of care to treat anemia due to CKD in dialysis.
•Presented a post-hoc analysis of all-cause mortality and hospitalization from the Phase 3 INNO2VATE trials of vadadustat at the American Society of Nephrology Kidney Week 2025 (ASN Kidney Week). The presentation entitled, “Win-Odds Analysis of Deaths and Hospitalization in Patients taking Vadadustat or Darbepoetin Alfa for CKD-Related Anemia Undergoing Dialysis,” demonstrated favorable and statistically significant effects of Vafseo relative to the ESA darbepoetin alfa on the hierarchical composite endpoint of death or hospitalization.
•Enrolled approximately 350 patients in VOCAL, a Phase IV trial evaluating TIW dosing of Vafseo versus ESAs, which is expected to report data in Q4 2026. The VOCAL trial also contains a sub-study of Vafseo’s impact on red blood cell characteristics.
Vafseo Q4 2025 Performance Expectations
•Total number of prescribers in Q4 was approximately 785, an increase of 8% over Q3.
•At least 25% of new patients in Q4 came from dialysis organizations other than U.S. Renal Care (USRC), an increase from less than 10% in Q3.
•We believe underlying patient dosing demand for Vafseo in Q4 2025 was between approximately $10.5 and $11.5 million.
•In Q4, we saw fewer patient starts at USRC centers anticipating the initiation of a new in-center observed dosing protocol and a decrease in average dose levels at centers that shifted to the observed dosing protocol. Within centers that adopted this protocol in Q4, first refill adherence rates improved from 75% in the first 9 months of 2025 to 91% in Q4. This new protocol at USRC contributed to an overall decrease in channel inventory of $4.5-$5 million. We expect Q4 2025 Vafseo net product revenue, driven in part by this inventory adjustment, will be in the range of $5-$6 million.
•We expect revenue growth to resume in Q1 2026 from increased patient access as well as anticipated improvement in adherence and compliance.
Rare Kidney Disease Pipeline Activities:
•Praliciguat is a soluble guanylate cyclase (sGC) stimulator being evaluated in a Phase 2 clinical trial for the treatment of biopsy-confirmed FSGS, a rare kidney disease, with plans to assess its use in other rare podocytopathies in the future. Akebia dosed the first patient in the FSGS study in December 2025. For more information about this study, please visit NCT07268638.
•AKB-097 (formerly known as ADX-097) is a tissue-targeted anti-C3d-Factor H fusion protein complement inhibitor that has potential applicability across a wide range of complement-mediated rare kidney diseases and is not expected to result in systemic complement inhibition seen with other inhibitors.
oAkebia plans to initiate an open label Phase 2 rare kidney disease basket study in the second half of 2026, with initial data generation expected in 2027.
oAkebia plans to evaluate IgA Nephropathy (IgAN), Lupus Nephritis (LN) and C3 Glomerulopathy (C3G) as part of the study.
Other Kidney Disease Pipeline Activities:
•AKB-9090 is a HIF-PH inhibitor that is entering Phase 1 for acute kidney injury associated with cardiac surgery in the first half of 2026. The drug candidate was shown to prevent kidney damage in an ischemia-reperfusion injury animal model.
About Akebia Therapeutics
Akebia Therapeutics, Inc. is a fully integrated biopharmaceutical company with the purpose to better the lives of people impacted by kidney disease. Akebia was founded in 2007 and is headquartered in Cambridge, Massachusetts. For more information, please visit our website at www.akebia.com, which does not form a part of this release.
Akebia Therapeutics, Inc. is a fully integrated biopharmaceutical company with the purpose to better the lives of people impacted by kidney disease. Akebia was founded in 2007 and is headquartered in Cambridge, Massachusetts. For more information, please visit our website at www.akebia.com, which does not form a part of this release.
About Vafseo® (vadadustat) tablets
Vafseo® (vadadustat) tablets is a once-daily oral hypoxia-inducible factor prolyl hydroxylase inhibitor that activates the physiologic response to hypoxia to stimulate endogenous production of erythropoietin, increasing hemoglobin and red blood cell production to manage anemia. Vafseo is approved for use in 37 countries.
Vafseo® (vadadustat) tablets is a once-daily oral hypoxia-inducible factor prolyl hydroxylase inhibitor that activates the physiologic response to hypoxia to stimulate endogenous production of erythropoietin, increasing hemoglobin and red blood cell production to manage anemia. Vafseo is approved for use in 37 countries.
INDICATION
In the Unites States, VAFSEO is indicated for the treatment of anemia due to chronic kidney disease (CKD) in adults who have been receiving dialysis for at least three months.
Limitations of Use
•VAFSEO has not been shown to improve quality of life, fatigue, or patient well-being.
•VAFSEO is not indicated for use:
•As a substitute for red blood cell transfusions in patients who require immediate correction of anemia.
•In patients with anemia due to CKD not on dialysis.
IMPORTANT SAFETY INFORMATION about VAFSEO (vadadustat) tablets
WARNING: INCREASED RISK OF DEATH, MYOCARDIAL INFARCTION, STROKE, VENOUS THROMBOEMBOLISM, and THROMBOSIS OF VASCULAR ACCESS.
VAFSEO increases the risk of thrombotic vascular events, including major adverse cardiovascular events (MACE).
Targeting a hemoglobin level greater than 11 g/dL is expected to further increase the risk of death and arterial and venous thrombotic events, as occurs with erythropoietin stimulating agents (ESAs), which also increase erythropoietin levels.
No trial has identified a hemoglobin target level, dose of VAFSEO, or dosing strategy that does not increase these risks.
Use the lowest dose of VAFSEO sufficient to reduce the need for red blood cell transfusions.
CONTRAINDICATIONS
•Known hypersensitivity to VAFSEO or any of its components
•Uncontrolled hypertension
WARNINGS AND PRECAUTIONS
•Increased Risk of Death, Myocardial Infarction (MI), Stroke, Venous Thromboembolism, and Thrombosis of Vascular Access
A rise in hemoglobin (Hb) levels greater than 1 g/dL over 2 weeks can increase these risks. Avoid in patients with a history of MI, cerebrovascular event, or acute coronary syndrome within the 3 months prior to starting VAFSEO. Targeting a Hb level of greater than 11 g/dL is expected to further increase the risk of death and arterial and venous thrombotic events. Use the lowest effective dose to reduce the need for red blood cell (RBC) transfusions. Adhere to dosing and Hb monitoring recommendations to avoid excessive erythropoiesis.
A rise in hemoglobin (Hb) levels greater than 1 g/dL over 2 weeks can increase these risks. Avoid in patients with a history of MI, cerebrovascular event, or acute coronary syndrome within the 3 months prior to starting VAFSEO. Targeting a Hb level of greater than 11 g/dL is expected to further increase the risk of death and arterial and venous thrombotic events. Use the lowest effective dose to reduce the need for red blood cell (RBC) transfusions. Adhere to dosing and Hb monitoring recommendations to avoid excessive erythropoiesis.
•Hepatotoxicity
Hepatocellular injury attributed to VAFSEO was reported in less than 1% of patients, including one severe case with jaundice. Elevated serum ALT, AST, and bilirubin levels were observed in 1.8%, 1.8%, and 0.3% of CKD patients treated with VAFSEO, respectively. Measure ALT, AST, and bilirubin before treatment and monthly for the first 6 months, then as clinically indicated. Discontinue VAFSEO if ALT or AST is persistently elevated or accompanied by elevated bilirubin. Not recommended in patients with cirrhosis or active, acute liver disease.
Hepatocellular injury attributed to VAFSEO was reported in less than 1% of patients, including one severe case with jaundice. Elevated serum ALT, AST, and bilirubin levels were observed in 1.8%, 1.8%, and 0.3% of CKD patients treated with VAFSEO, respectively. Measure ALT, AST, and bilirubin before treatment and monthly for the first 6 months, then as clinically indicated. Discontinue VAFSEO if ALT or AST is persistently elevated or accompanied by elevated bilirubin. Not recommended in patients with cirrhosis or active, acute liver disease.
•Hypertension
Worsening of hypertension was reported in 14% of VAFSEO and 17% of darbepoetin alfa patients. Serious worsening of hypertension was reported in 2.7% of VAFSEO and 3% of darbepoetin alfa patients. Cases of hypertensive crisis, including hypertensive encephalopathy and seizures, have also been reported in patients receiving VAFSEO. Monitor blood pressure. Adjust anti-hypertensive therapy as needed.
Worsening of hypertension was reported in 14% of VAFSEO and 17% of darbepoetin alfa patients. Serious worsening of hypertension was reported in 2.7% of VAFSEO and 3% of darbepoetin alfa patients. Cases of hypertensive crisis, including hypertensive encephalopathy and seizures, have also been reported in patients receiving VAFSEO. Monitor blood pressure. Adjust anti-hypertensive therapy as needed.
•Seizures
Seizures occurred in 1.6% of VAFSEO and 1.6% of darbepoetin alfa patients. Monitor for new-onset seizures, premonitory symptoms, or change in seizure frequency.
Seizures occurred in 1.6% of VAFSEO and 1.6% of darbepoetin alfa patients. Monitor for new-onset seizures, premonitory symptoms, or change in seizure frequency.
•Gastrointestinal (GI) Erosion
Gastric or esophageal erosions occurred in 6.4% of VAFSEO and 5.3% of darbepoetin alfa
Gastric or esophageal erosions occurred in 6.4% of VAFSEO and 5.3% of darbepoetin alfa
patients. Serious GI erosions, including GI bleeding and the need for RBC transfusions, were reported in 3.4% of VAFSEO and 3.3% of darbepoetin alfa patients. Consider this risk in patients at increased risk of GI erosion. Advise patients about signs of erosions and GI bleeding and urge them to seek prompt medical care if present.
•Serious Adverse Reactions in Patients with Anemia Due to CKD and Not on Dialysis
The safety of VAFSEO has not been established for the treatment of anemia due to CKD in adults not on dialysis and its use is not recommended in this setting. In large clinical trials in adults with anemia of CKD who were not on dialysis, an increased risk of mortality, stroke, MI, serious acute kidney injury, serious hepatic injury, and serious GI erosions was observed in patients treated with VAFSEO compared to darbepoetin alfa.
The safety of VAFSEO has not been established for the treatment of anemia due to CKD in adults not on dialysis and its use is not recommended in this setting. In large clinical trials in adults with anemia of CKD who were not on dialysis, an increased risk of mortality, stroke, MI, serious acute kidney injury, serious hepatic injury, and serious GI erosions was observed in patients treated with VAFSEO compared to darbepoetin alfa.
•Malignancy
VAFSEO has not been studied and is not recommended in patients with active malignancies. Malignancies were observed in 2.2% of VAFSEO and 3.0% of darbepoetin alfa patients. No evidence of increased carcinogenicity was observed in animal studies.
VAFSEO has not been studied and is not recommended in patients with active malignancies. Malignancies were observed in 2.2% of VAFSEO and 3.0% of darbepoetin alfa patients. No evidence of increased carcinogenicity was observed in animal studies.
ADVERSE REACTIONS
•The most common adverse reactions (occurring at ≥ 10%) were hypertension and diarrhea.
DRUG INTERACTIONS
•Iron supplements and iron-containing phosphate binders: Administer VAFSEO at least 1 hour before products containing iron.
•Non-iron-containing phosphate binders: Administer VAFSEO at least 1 hour before or 2 hours after non-iron-containing phosphate binders.
•BCRP substrates: Monitor for signs of substrate adverse reactions and consider dose reduction.
•Statins: Monitor for statin-related adverse reactions. Limit the daily dose of simvastatin to 20 mg and rosuvastatin to 5 mg.
USE IN SPECIFIC POPULATIONS
•Pregnancy: May cause fetal harm.
•Lactation: Breastfeeding not recommended until two days after the final dose.
•Hepatic Impairment: Not recommended in patients with cirrhosis or active, acute liver disease.
Please note that this information is not comprehensive. Please click here for the Full Prescribing Information, including BOXED WARNING and Medication Guide.
Forward-Looking Statements
Statements in this press release regarding Akebia Therapeutics, Inc.'s ("Akebia's") strategy, plans, prospects, expectations, beliefs, intentions and goals are forward-looking statements
Statements in this press release regarding Akebia Therapeutics, Inc.'s ("Akebia's") strategy, plans, prospects, expectations, beliefs, intentions and goals are forward-looking statements
within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, as amended, and include, but are not limited to, statements regarding: Akebia’s expectations and beliefs about demand for Vafseo in 2026, including the number of patients with access to Vafseo and that demand and depth of prescribing for Vafseo will increase in 2026; Akebia’s beliefs and expectations with respect to its financial position; Akebia’s beliefs that existing customers will accelerate adoption of Vafseo and new customers will operationalize Vafseo protocols within their organizations; Akebia’s plans to generate post-marketing Vafseo clinical data that will support its goal to make Vafseo standard of care to treat anemia due to CKD in dialysis; Akebia’s plans and expectations with respect to aggressively progressing its pipeline and leveraging its scientific leadership in nephrology with an aim to help patients in need of new therapies; Akebia’s beliefs that revenue-generating products are the engine driving advancement of its mid-stage pipeline, which, along with continued adoption of Vafseo, can drive significant shareholder value this year and beyond; Akebia’s beliefs with respect to patient dosing demand for Vafseo in Q4 2025, including impacts from a new observed dosing protocol at USRC; Akebia’s expectations with respect to Q4 2025 Vafseo net product revenue and that revenue growth will resume in Q1 2026 from increased patient access and anticipated improvement in adherence and compliance; Akebia’s plans and expectations with respect to the VOICE trial, including the timing of topline results and potential to help establish Vafseo as a standard of care to treat anemia due to CKD in dialysis; Akebia’s plans and expectations with respect to the VOCAL trial, including timing of data; Akebia’s plans to assess the use of praliciguat in other rare podocytopathies; Akebia’s plans and expectations with respect AKB-097, including the timing of initiation of, and initial data from, an open label Phase 2 basket study and the indications to be evaluated; and Akebia’s plans and expectations with respect to AKB-9090, including the timing of a Phase 1 trial.
The terms "intend," "believe," "plan," "goal," "potential," "anticipate, "estimate," "expect," "future," "will," "continue," derivatives of these words, and similar references are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Actual results, performance or experience may differ materially from those expressed or implied by any forward-looking statement as a result of various risks, uncertainties and other factors, including, but not limited to, risks associated with: the potential therapeutic benefits, safety profile, and effectiveness of Vafseo and Akebia’s development candidates; the results of preclinical and clinical research; Akebia’s ability to initiate and enroll patients in its clinical trials; decisions made by health authorities, such as the FDA, with respect to regulatory filings and other interactions; the potential demand and market potential and acceptance of, as well as coverage and reimbursement related to, Vafseo®, including estimates regarding the potential market opportunity; the competitive landscape for Auryxia® and Vafseo, including generic entrants and the timing thereof; the ability of Akebia to attract and retain qualified personnel; Akebia's ability to achieve and maintain profitability and to maintain operating expenses consistent with its operating plan; manufacturing, supply chain and quality matters and any recalls, write-downs, impairments or other related consequences or potential consequences; early termination of any of Akebia's collaborations; and changes in the geopolitical environment and uncertainty surrounding U.S. trade policy on tariffs. Other risks and uncertainties include those identified under the heading "Risk Factors" in Akebia's Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, and other filings
that Akebia may make with the U.S. Securities and Exchange Commission in the future. These forward-looking statements (except as otherwise noted) speak only as of the date of this press release, and, except as required by law, Akebia does not undertake, and specifically disclaims, any obligation to update any forward-looking statements contained in this press release.
Akebia Therapeutics®, Auryxia® and Vafseo® are registered trademarks of Akebia Therapeutics, Inc. and its affiliates.
Akebia Therapeutics Contact
Mercedes Carrasco
mcarrasco@akebia.com
Mercedes Carrasco
mcarrasco@akebia.com

1 Bettering the Lives of People Impacted by Kidney DiseaseCorporate Presentation January 2026 NASDAQ: AKBA JOHN BUTLER Chief Executive Officer

Cautionary note on forward-looking statements Statements in this presentation regarding Akebia Therapeutics, Inc.’s (“Akebia’s”) strategy, plans, prospects, expectations, beliefs, intentions and goals are forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, as amended, and include, but are not limited to, statements regarding: Akebia's plans, strategies and prospects for its business; Akebia’s plans with respect to its U.S. commercial launch of Vafseo®, including the potential U.S. market opportunity; Akebia’s plans for Vafseo to become standard of care for treatment of anemia due to CKD in dialysis, including its ability to build on the body of evidence demonstrating Vafseo’s value potential, and progress towards that goal; Akebia’s expectations and beliefs about demand for Vafseo, including the number of patients with access to Vafseo and the focus of dialysis organizations; Akebia’s beliefs with respect to patient dosing demand for Vafseo in 2026; Akebia’s plans and expectations with respect to publication of additional analyses of INNO2VATE data; Akebia’s plans and expectations with respect to the VOICE trial, including the timing of top-line data and potential to demonstrate favorable outcomes in the composite of all-cause mortality and hospitalization in patients treated with vadadustat compared to ESA; Akebia’s plans and expectations with respect to the VOCAL trial, including timing of top-line data; Akebia’s plans and expectations with respect to the praliciguat trial, including to assess the use of praliciguat in other rare podocytopathies, the number of patients be enrolled in the trial and its potential for successful development and regulatory path; Akebia’s plans and expectations with respect AKB-097, including the timing of initiation of, and initial data from, an open label Phase 2 basket study and the indications to be evaluated, other potential indications for consideration and its potential for pipeline in a product and to achieve opportunities to address unmet need; and Akebia’s plans and expectations with respect to AKB-9090, including the timing of initiation of, and top-line data from, a Phase 1 trial and the indication to be evaluated. The terms "intend," "believe," "plan," "goal," "potential," "anticipate, "estimate," "expect," "future," "will," "continue," derivatives of these words, and similar references are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Actual results, performance or experience may differ materially from those expressed or implied by any forward-looking statement as a result of various risks, uncertainties and other factors, including, but not limited to, risks associated with: the potential therapeutic benefits, safety profile, and effectiveness of Vafseo and Akebia’s development candidates; the results of preclinical and clinical research; Akebia’s ability to initiate and enroll patients in its clinical trials; decisions made by health authorities, such as the FDA, with respect to regulatory filings and other interactions; the potential demand and market potential and acceptance of, as well as coverage and reimbursement related to, Vafseo®, including estimates regarding the potential market opportunity; the competitive landscape for Auryxia® and Vafseo, including generic entrants and the timing thereof; the ability of Akebia to attract and retain qualified personnel; Akebia's ability to achieve and maintain profitability and to maintain operating expenses consistent with its operating plan; manufacturing, supply chain and quality matters and any recalls, write-downs, impairments or other related consequences or potential consequences; early termination of any of Akebia's collaborations; and changes in the geopolitical environment and uncertainty surrounding U.S. trade policy on tariffs. Other risks and uncertainties include those identified under the heading "Risk Factors" in Akebia's Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, and other filings that Akebia may make with the U.S. Securities and Exchange Commission in the future. These forward-looking statements (except as otherwise noted) speak only as of the date of this presentation, and, except as required by law, Akebia does not undertake, and specifically disclaims, any obligation to update any forward-looking statements contained in this presentation. Akebia Therapeutics®, Auryxia® and Vafseo® are registered trademarks of Akebia Therapeutics, Inc. and its affiliates. 2
Purpose-driven integrated biotechnology company focused on kidney disease 3 Experienced team leveraging expertise in kidney disease and relationships with patient community and key medical experts Two commercial drugs addressing large markets within kidney disease Mid-stage pipeline targeting multiple rare kidney diseases Early-stage pipeline including kidney and non-kidney indications
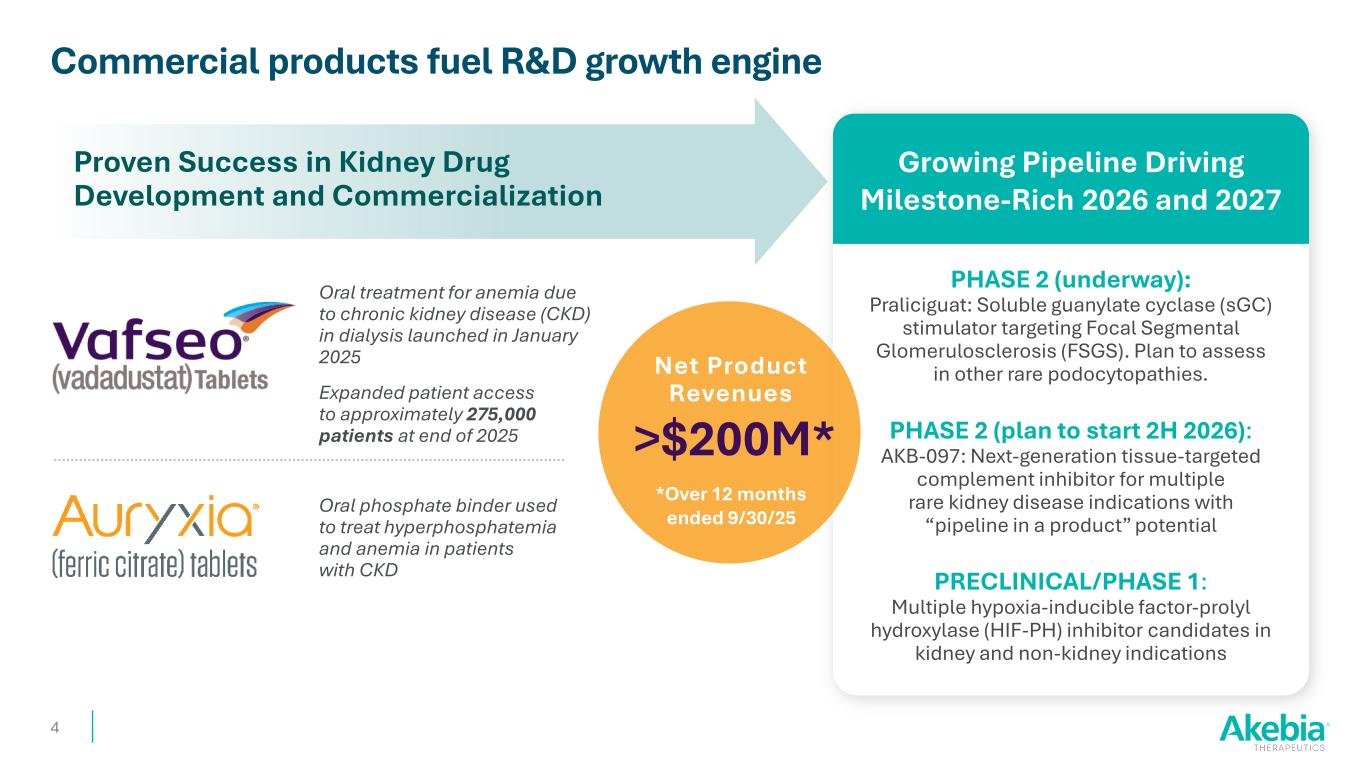
Commercial products fuel R&D growth engine Proven Success in Kidney Drug Development and Commercialization Growing Pipeline Driving Milestone-Rich 2026 and 2027 Oral phosphate binder used to treat hyperphosphatemia and anemia in patients with CKD Oral treatment for anemia due to chronic kidney disease (CKD) in dialysis launched in January 2025 Expanded patient access to approximately 275,000 patients at end of 2025 PHASE 2 (underway): Praliciguat: Soluble guanylate cyclase (sGC) stimulator targeting Focal Segmental Glomerulosclerosis (FSGS). Plan to assess in other rare podocytopathies. PHASE 2 (plan to start 2H 2026): AKB-097: Next-generation tissue-targeted complement inhibitor for multiple rare kidney disease indications with “pipeline in a product” potential PRECLINICAL/PHASE 1: Multiple hypoxia-inducible factor-prolyl hydroxylase (HIF-PH) inhibitor candidates in kidney and non-kidney indications 4 Net Product Revenues >$200M* *Over 12 months ended 9/30/25
Vafseo® (vadadustat) Tablets indicated for the treatment of anemia due to CKD in adults who have been receiving dialysis for at least three months Click here for the Full Prescribing Information, including BOXED WARNING and Medication Guide. Foundational Launch Year Sets the Stage for Goal to Become Standard of Care 5 This presentation is intended for investor purposes only and is not intended for promotional purposes

An oral HIF-PH inhibitor 6 Unique mechanism of action built on Nobel Prize-winning science Stimulates body’s natural response to hypoxia Enhances body’s natural production of EPO Activates iron mobilization Simple titration and fewer dose modifications Convenient oral dosing Controls hemoglobin (Hb) levels over time

Dialysis organization protocols in place allowing prescribing access for ~275,000 patients Initiated additional clinical trials Davita completed operational pilot 7 Milestones point to Vafseo potential to become standard of care in $1 billion market1 • DOs initiate three times weekly (TIW) dosing regimen and focus on home dialysis patients • Publication of win-odds analysis of INNO2VATE data expected • VOCAL top-line readout in Q4 2026 2026 TDAPA YEAR 2 Positioned for Inflection PATH TO STANDARD OF CARE 2025 TDAPA YEAR 1 Foundational Launch • Vafseo enters the CMS bundle in January 2027 • $1 billion market opportunity at current erythropoiesis-stimulating agent (ESA) pricing • VOICE outcomes study top-line read out expected in Q1 2027 with potential to demonstrate favorable outcomes in the composite of all-cause mortality and hospitalization in patients treated with vadadustat compared to ESA 2027 and beyond Poised to Capture Significant Market Share

Building on body of evidence demonstrating Vafseo’s value proposition VOCAL 8 Patients randomized to oral Vafseo 300 mg tablets administered TIW or ESA WIN-ODDS ANALYSIS Post-hoc analysis of completed Phase 3 INNO2VATE clinical trials Demonstrated favorable and statistically significant effects of Vafseo relative to the erythropoiesis-stimulating agent (ESA) darbepoetin alfa on the composite endpoint of death and hospitalization2 Includes a sub-study evaluating impact of Vafseo on red blood cell characteristics ~350 Patients enrolled Top-line data expected Q4 2026 VOICE Patients randomized to oral Vafseo 300 mg tablets administered TIW or ESA Evaluating Vafseo vs ESA using hierarchical endpoint of all-cause mortality and all-cause hospitalization 2,116 Patients enrolled Top-line data expected early 2027
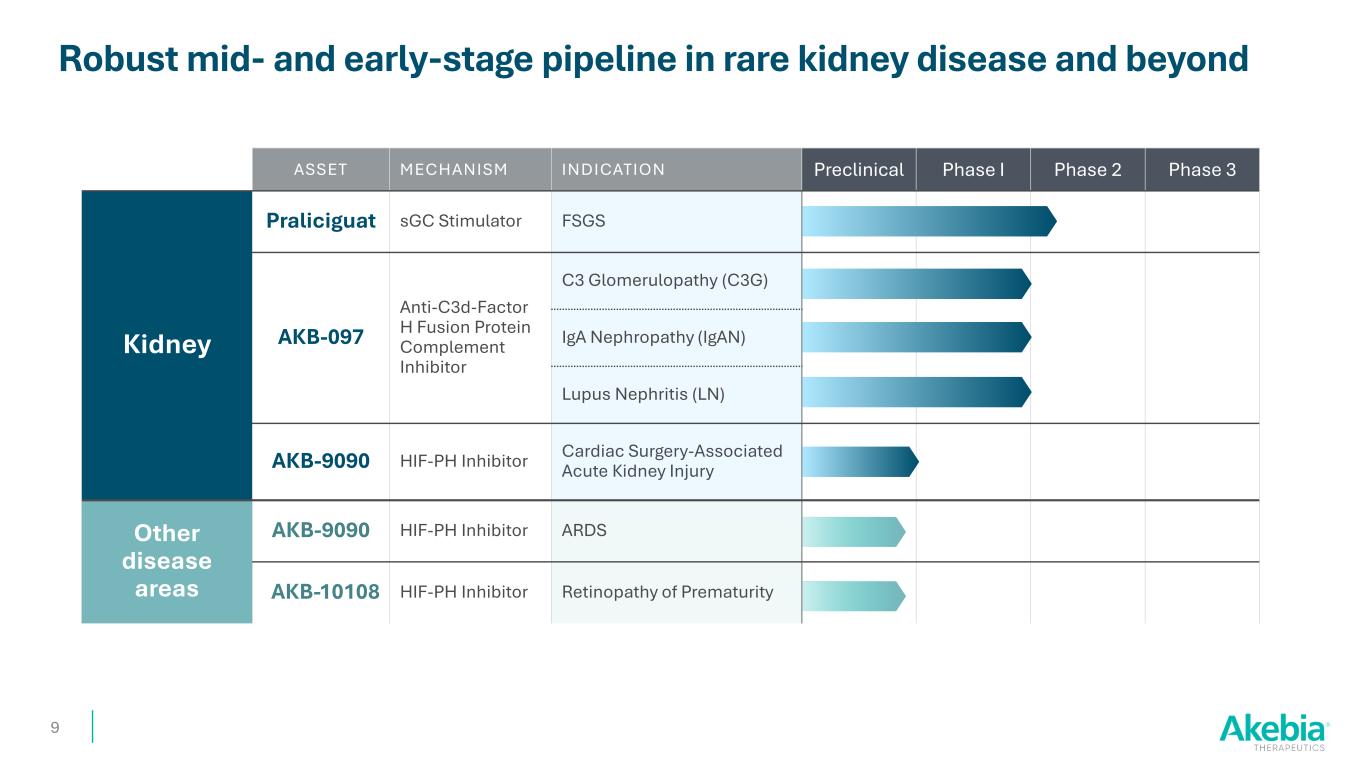
ASSET MECHANISM INDICATION Preclinical Phase I Phase 2 Phase 3 Kidney Praliciguat sGC Stimulator FSGS AKB-097 Anti-C3d-Factor H Fusion Protein Complement Inhibitor C3 Glomerulopathy (C3G) IgA Nephropathy (IgAN) Lupus Nephritis (LN) AKB-9090 HIF-PH Inhibitor Cardiac Surgery-Associated Acute Kidney Injury Other disease areas AKB-9090 HIF-PH Inhibitor ARDS AKB-10108 HIF-PH Inhibitor Retinopathy of Prematurity Robust mid- and early-stage pipeline in rare kidney disease and beyond 9
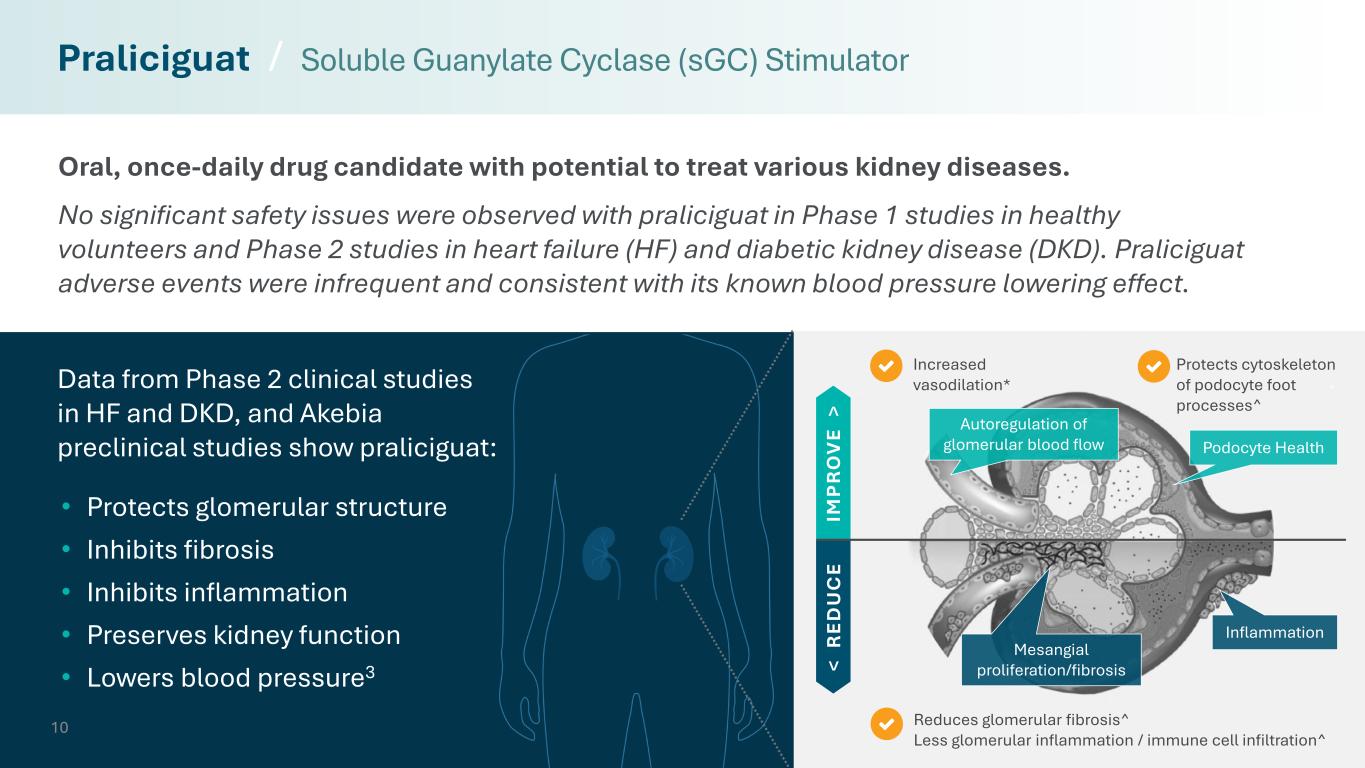
10 Reduces glomerular fibrosis^ Less glomerular inflammation / immune cell infiltration^ Protects cytoskeleton of podocyte foot processes^ Increased vasodilation* Oral, once-daily drug candidate with potential to treat various kidney diseases. No significant safety issues were observed with praliciguat in Phase 1 studies in healthy volunteers and Phase 2 studies in heart failure (HF) and diabetic kidney disease (DKD). Praliciguat adverse events were infrequent and consistent with its known blood pressure lowering effect. Praliciguat / Soluble Guanylate Cyclase (sGC) Stimulator < R ED U C E IM P R O V E > Inflammation Mesangial proliferation/fibrosis • Protects glomerular structure • Inhibits fibrosis • Inhibits inflammation • Preserves kidney function • Lowers blood pressure3 Autoregulation of glomerular blood flow Podocyte Health Data from Phase 2 clinical studies in HF and DKD, and Akebia preclinical studies show praliciguat:
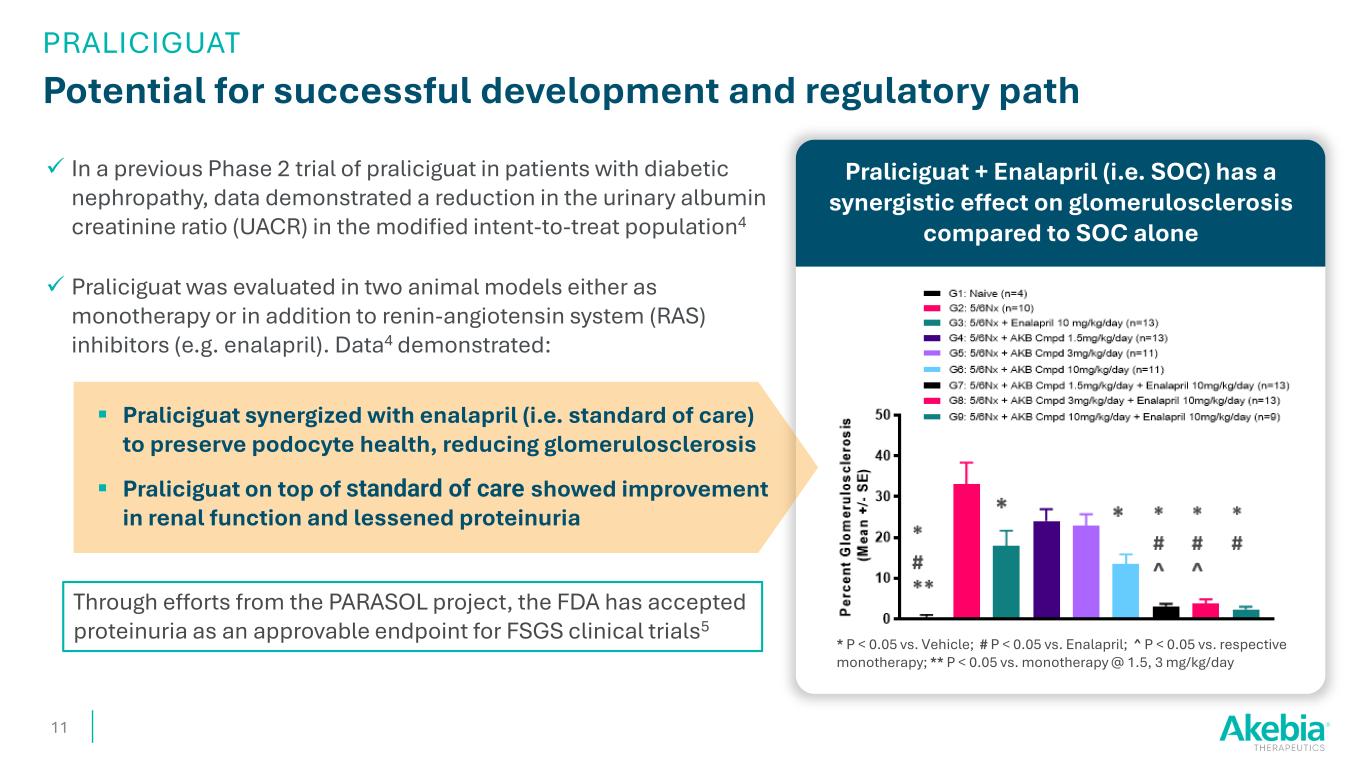
In a previous Phase 2 trial of praliciguat in patients with diabetic nephropathy, data demonstrated a reduction in the urinary albumin creatinine ratio (UACR) in the modified intent-to-treat population4 Praliciguat was evaluated in two animal models either as monotherapy or in addition to renin-angiotensin system (RAS) inhibitors (e.g. enalapril). Data4 demonstrated: Praliciguat synergized with enalapril (i.e. standard of care) to preserve podocyte health, reducing glomerulosclerosis Praliciguat on top of standard of care showed improvement in renal function and lessened proteinuria Praliciguat + Enalapril (i.e. SOC) has a synergistic effect on glomerulosclerosis compared to SOC alone Potential for successful development and regulatory path 11 * P < 0.05 vs. Vehicle; # P < 0.05 vs. Enalapril; ^ P < 0.05 vs. respective monotherapy; ** P < 0.05 vs. monotherapy @ 1.5, 3 mg/kg/day Through efforts from the PARASOL project, the FDA has accepted proteinuria as an approvable endpoint for FSGS clinical trials5 PRALICIGUAT
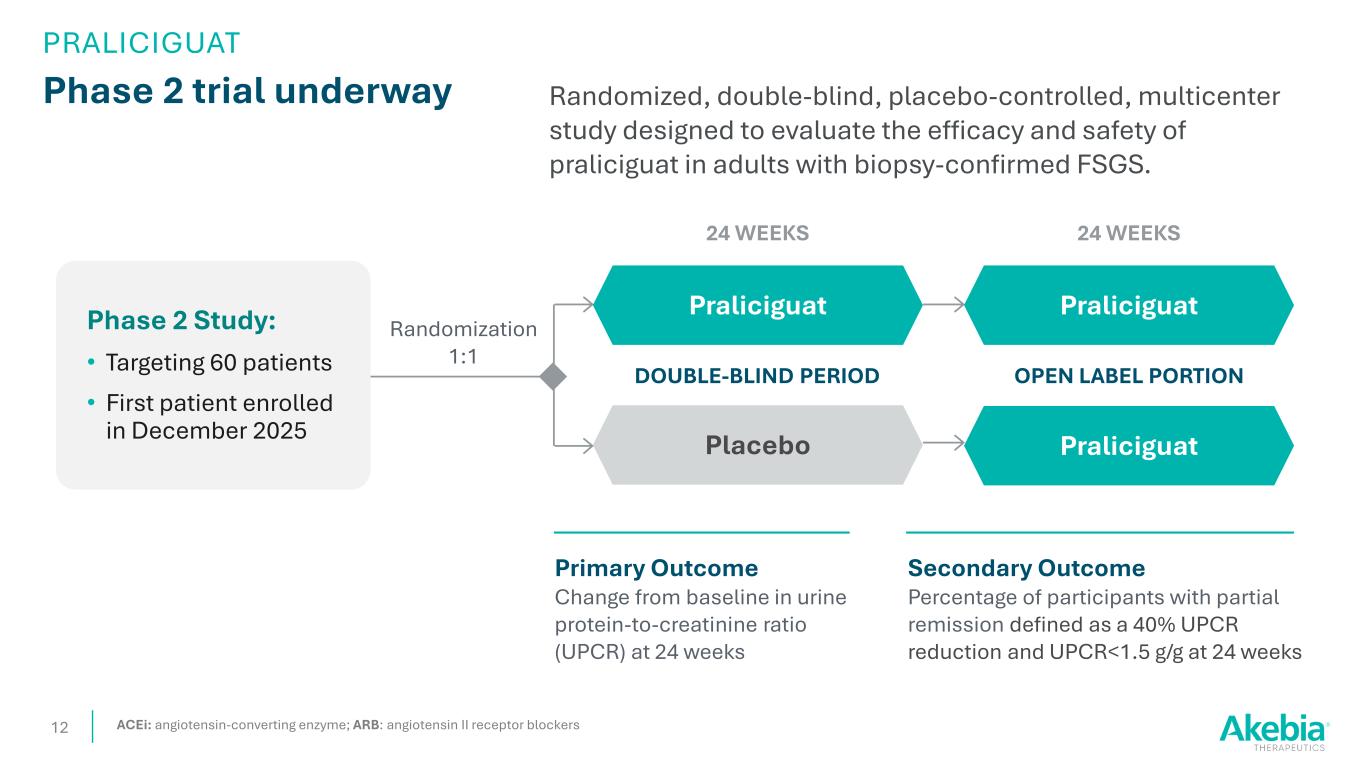
Phase 2 trial underway 12 Randomized, double-blind, placebo-controlled, multicenter study designed to evaluate the efficacy and safety of praliciguat in adults with biopsy-confirmed FSGS. Randomization 1:1 Placebo Praliciguat Secondary Outcome Percentage of participants with partial remission defined as a 40% UPCR reduction and UPCR<1.5 g/g at 24 weeks DOUBLE-BLIND PERIOD Primary Outcome Change from baseline in urine protein-to-creatinine ratio (UPCR) at 24 weeks 24 WEEKS OPEN LABEL PORTION Praliciguat Praliciguat 24 WEEKS ACEi: angiotensin-converting enzyme; ARB: angiotensin II receptor blockers PRALICIGUAT Phase 2 Study: • Targeting 60 patients • First patient enrolled in December 2025

Targeting unmet needs in FSGS FSGS can cause various symptoms including high blood pressure, proteinuria and kidney failure • A leading glomerular cause of end-stage kidney disease in the U.S.8 • Accounts for 40% of adults with severe proteinuria (nephrotic syndrome)9 13 FSGS is a condition characterized by focal and segmental scarring in the kidney’s filtering units known as glomeruli. 40,000 people in the U.S.6 No treatments are specifically indicated for FSGS; current treatments such as steroids, other immunosuppressives, and antihypertensives may slow kidney failure progression in some patients. 220,000 people worldwide7 DIAGNOSED IN APPROXIMATELY PRALICIGUAT
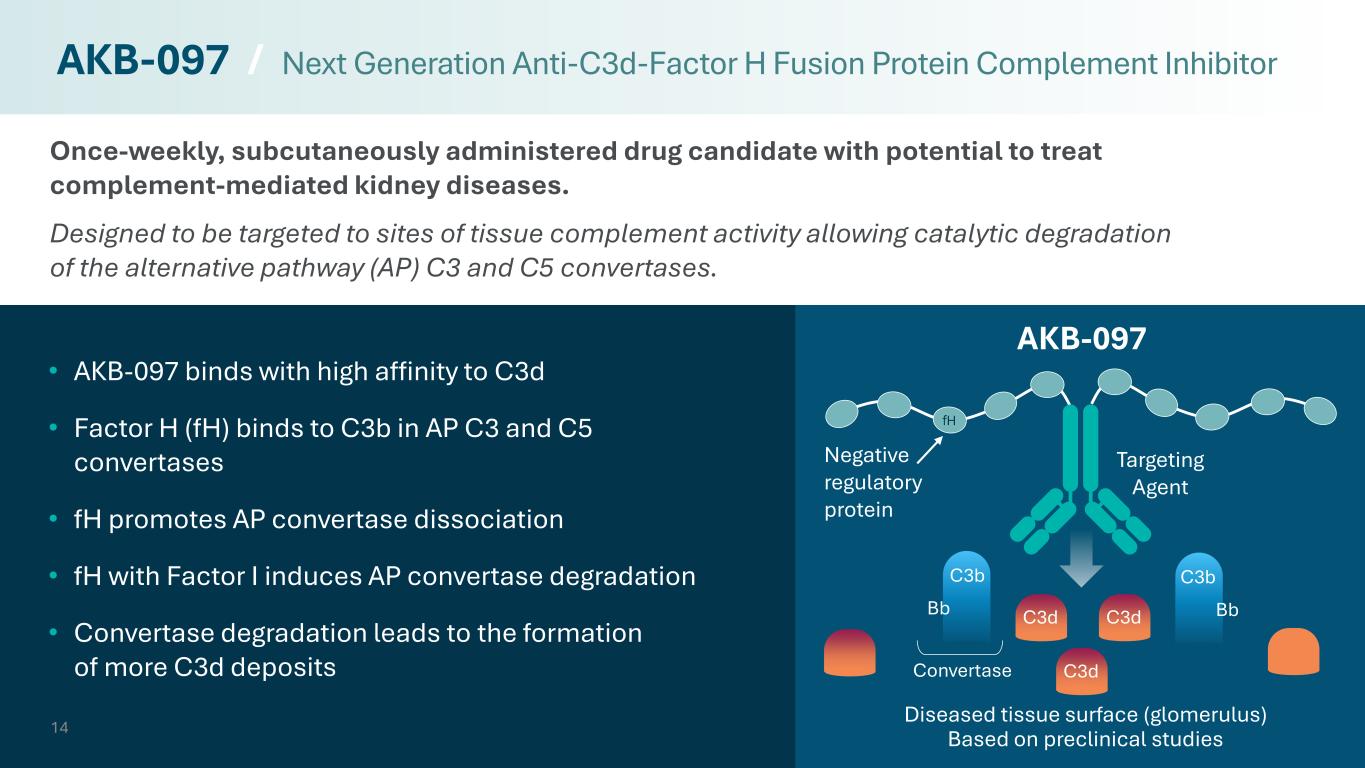
Once-weekly, subcutaneously administered drug candidate with potential to treat complement-mediated kidney diseases. Designed to be targeted to sites of tissue complement activity allowing catalytic degradation of the alternative pathway (AP) C3 and C5 convertases. 14 • AKB-097 binds with high affinity to C3d • Factor H (fH) binds to C3b in AP C3 and C5 convertases • fH promotes AP convertase dissociation • fH with Factor I induces AP convertase degradation • Convertase degradation leads to the formation of more C3d deposits AKB-097 / Next Generation Anti-C3d-Factor H Fusion Protein Complement Inhibitor C3b Bb C3b Bb Convertase C3d C3d C3d Targeting Agent Negative regulatory protein AKB-097 Diseased tissue surface (glomerulus) Based on preclinical studies fH
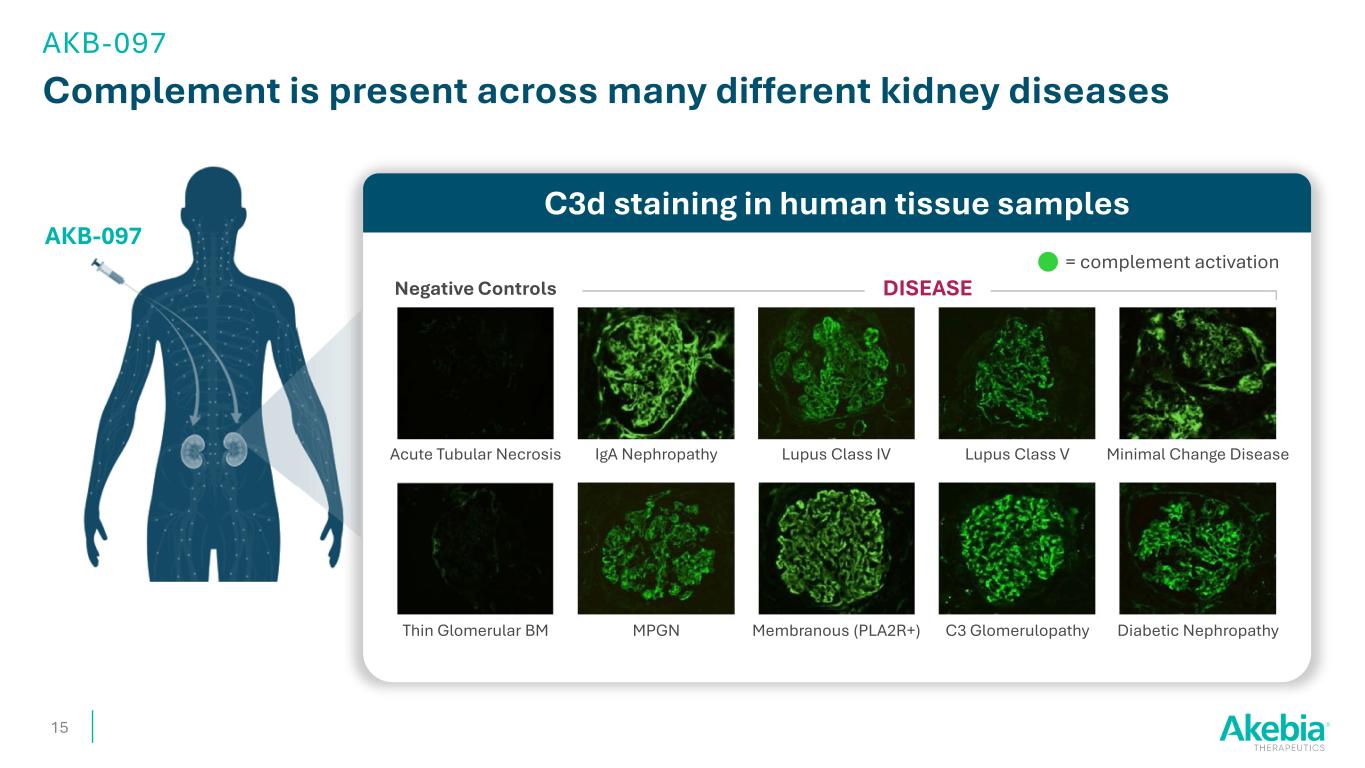
Complement is present across many different kidney diseases 15 AKB-097 C3d staining in human tissue samples DISEASENegative Controls Acute Tubular Necrosis Thin Glomerular BM MPGN Membranous (PLA2R+) C3 Glomerulopathy Diabetic Nephropathy Minimal Change DiseaseLupus Class VLupus Class IVIgA Nephropathy = complement activation AKB-097
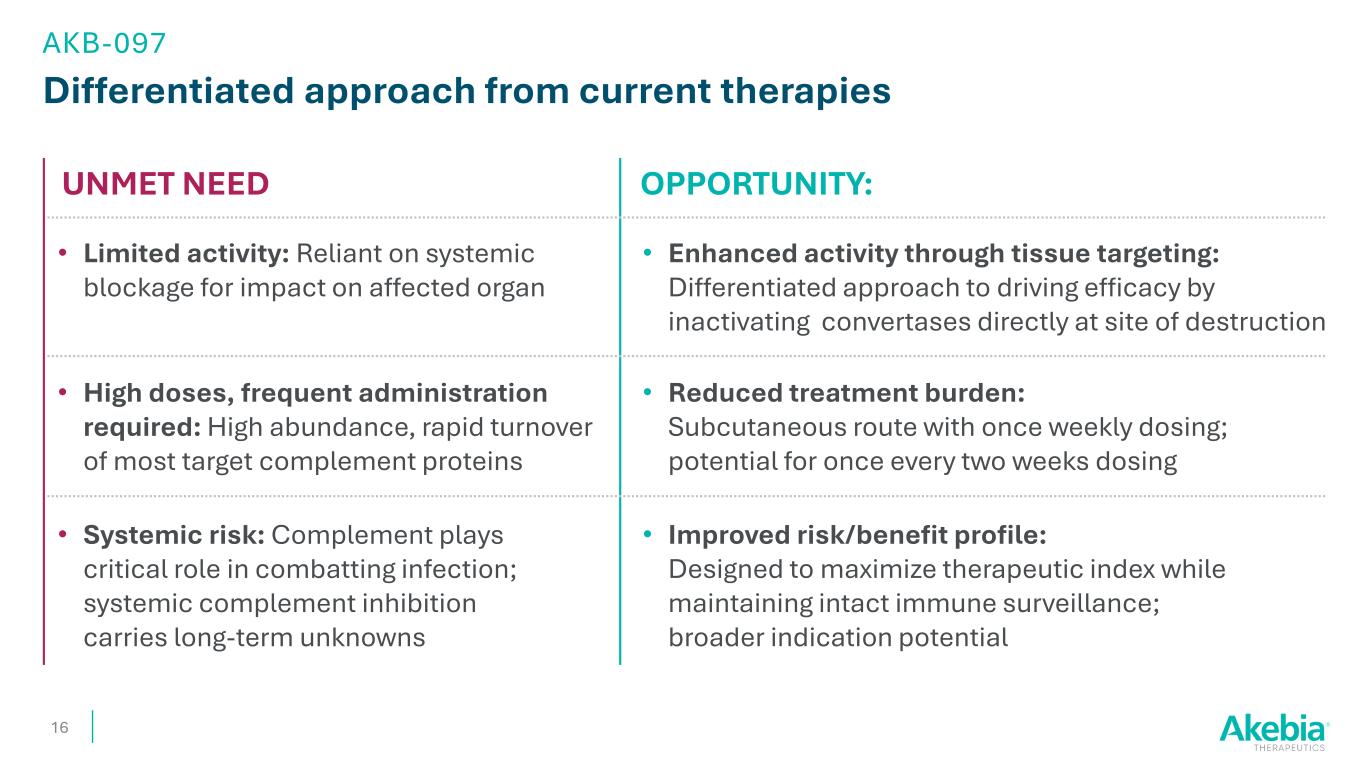
Differentiated approach from current therapies OPPORTUNITY: 16 • Enhanced activity through tissue targeting: Differentiated approach to driving efficacy by inactivating convertases directly at site of destruction • Limited activity: Reliant on systemic blockage for impact on affected organ UNMET NEED • Improved risk/benefit profile: Designed to maximize therapeutic index while maintaining intact immune surveillance; broader indication potential • Systemic risk: Complement plays critical role in combatting infection; systemic complement inhibition carries long-term unknowns • Reduced treatment burden: Subcutaneous route with once weekly dosing; potential for once every two weeks dosing • High doses, frequent administration required: High abundance, rapid turnover of most target complement proteins AKB-097
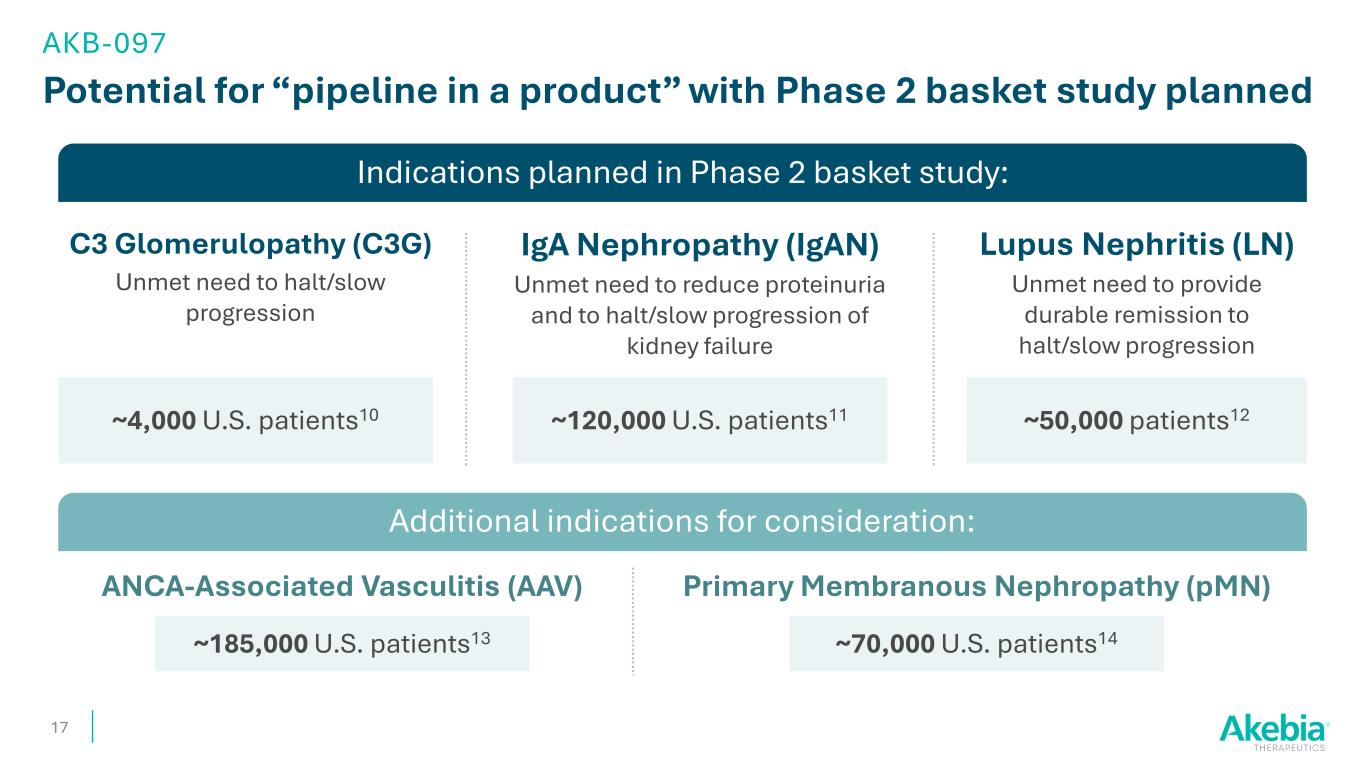
Indications planned in Phase 2 basket study: Potential for “pipeline in a product” with Phase 2 basket study planned 17 AKB-097 C3 Glomerulopathy (C3G) Unmet need to halt/slow progression IgA Nephropathy (IgAN) Unmet need to reduce proteinuria and to halt/slow progression of kidney failure Lupus Nephritis (LN) Unmet need to provide durable remission to halt/slow progression Additional indications for consideration: ~4,000 U.S. patients10 ~50,000 patients12 ANCA-Associated Vasculitis (AAV) Primary Membranous Nephropathy (pMN) ~185,000 U.S. patients13 ~70,000 U.S. patients14 ~120,000 U.S. patients11
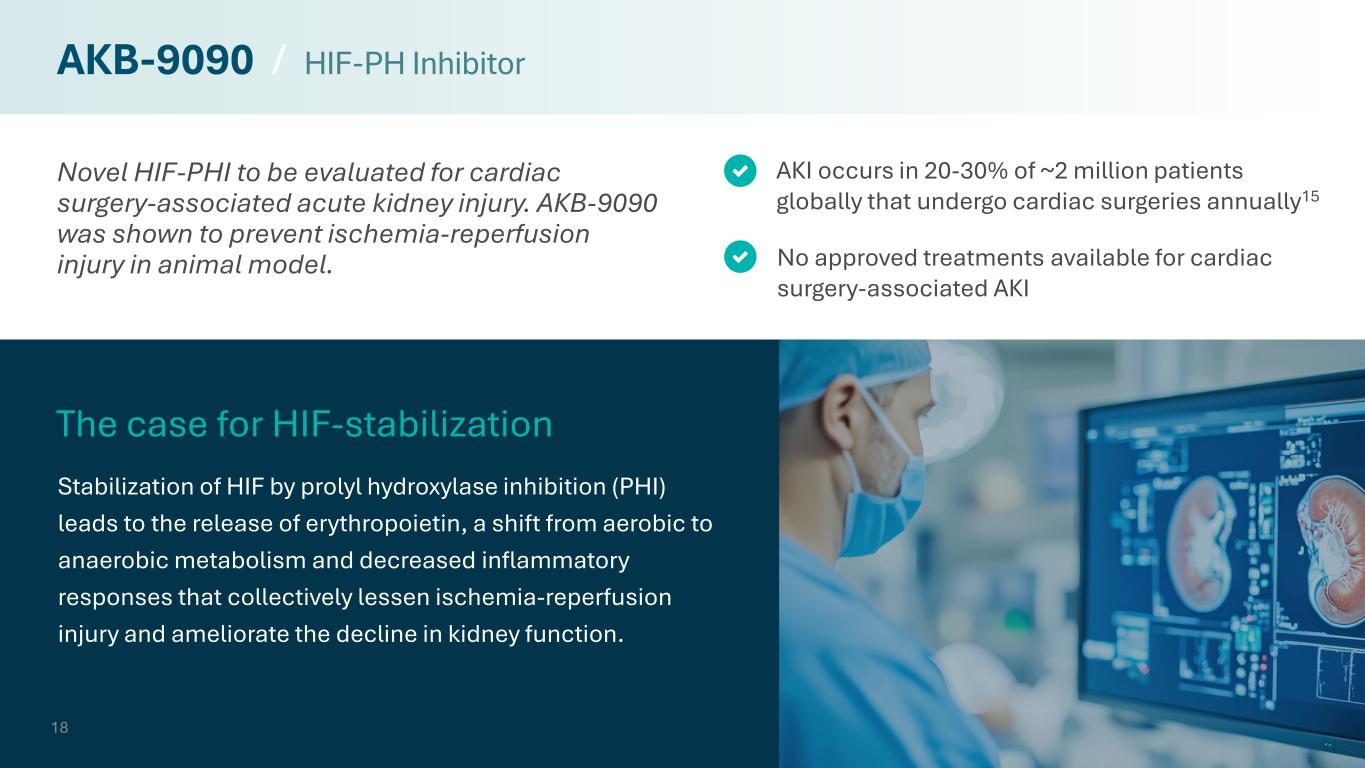
Novel HIF-PHI to be evaluated for cardiac surgery-associated acute kidney injury. AKB-9090 was shown to prevent ischemia-reperfusion injury in animal model. Stabilization of HIF by prolyl hydroxylase inhibition (PHI) leads to the release of erythropoietin, a shift from aerobic to anaerobic metabolism and decreased inflammatory responses that collectively lessen ischemia-reperfusion injury and ameliorate the decline in kidney function. No approved treatments available for cardiac surgery-associated AKI AKI occurs in 20-30% of ~2 million patients globally that undergo cardiac surgeries annually15 The case for HIF-stabilization 18 AKB-9090 / HIF-PH Inhibitor
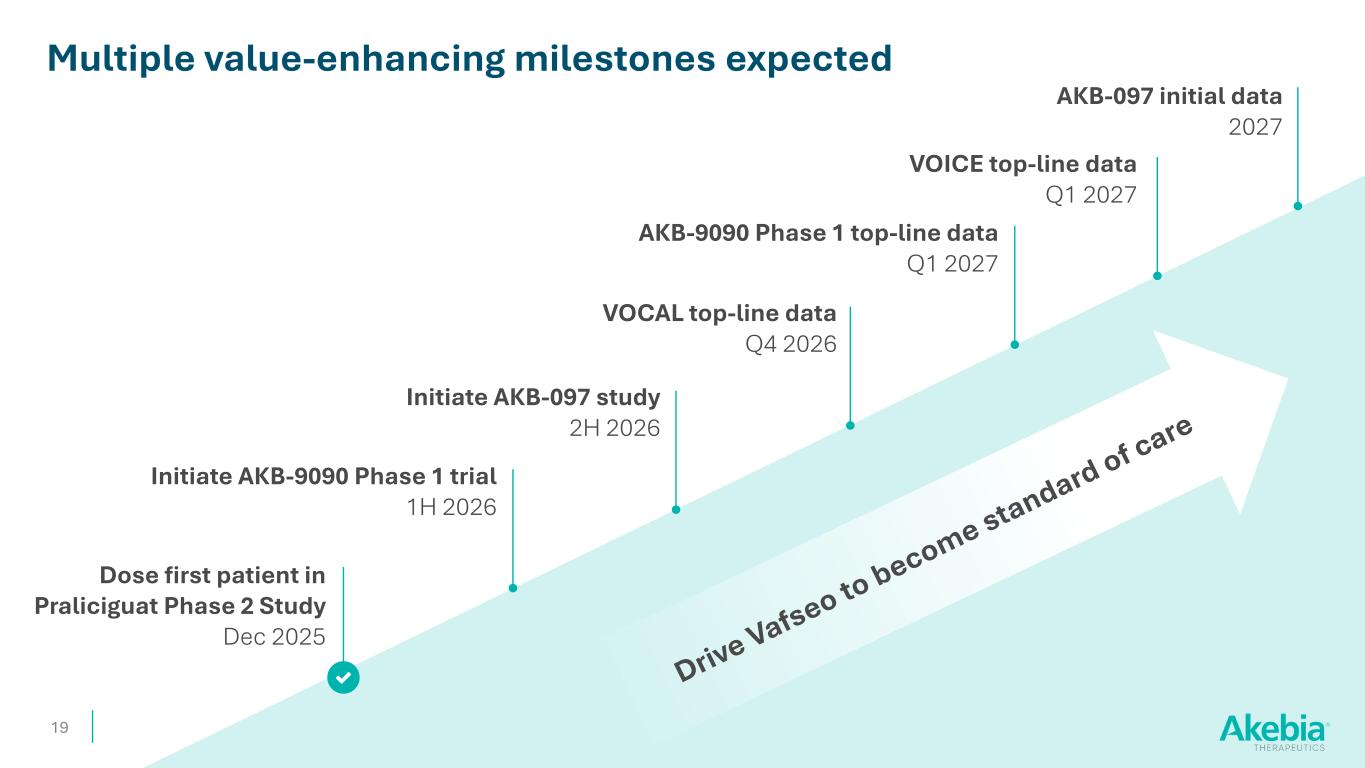
19 Dose first patient in Praliciguat Phase 2 Study Dec 2025 Multiple value-enhancing milestones expected Initiate AKB-9090 Phase 1 trial 1H 2026 VOCAL top-line data Q4 2026 Initiate AKB-097 study 2H 2026 AKB-097 initial data 2027 VOICE top-line data Q1 2027 AKB-9090 Phase 1 top-line data Q1 2027

Akebia: A Compelling Investment Opportunity in the Kidney Space 20 • Two FDA-approved, revenue-generating products; supported by an experienced commercial organization • Potential for Vafseo to be standard of care for treatment of anemia due to CKD in dialysis; $1 billion U.S. market opportunity1 • Advancing differentiated mid-stage pipeline in rare kidney disease • Strong balance sheet; $166 million in cash & cash equivalents as of 9/30/2025 • Multiple value-enhancing milestones expected in 2026 & 2027

21 Appendix

WARNING: INCREASED RISK OF DEATH, MYOCARDIAL INFARCTION, STROKE, VENOUS THROMBOEMBOLISM, and THROMBOSIS OF VASCULAR ACCESS. See full prescribing information for complete boxed warning. VAFSEO increases the risk of thrombotic vascular events, including major adverse cardiovascular events (MACE). Targeting a hemoglobin level greater than 11 g/dL is expected to further increase the risk of death and arterial and venous thrombotic events, as occurs with erythropoietin stimulating agents (ESAs), which also increase erythropoietin levels. No trial has identified a hemoglobin target level, dose of VAFSEO, or dosing strategy that does not increase these risks. Use the lowest dose of VAFSEO sufficient to reduce the need for red blood cell transfusions. IMPORTANT SAFETY INFORMATION about VAFSEO (vadadustat) tablets 22

CONTRAINDICATIONS • Known hypersensitivity to VAFSEO or any of its components • Uncontrolled hypertension WARNINGS AND PRECAUTIONS Increased Risk of Death, Myocardial Infarction, Stroke, Venous Thromboembolism, and Thrombosis of Vascular Access A rise in hemoglobin (Hb) levels greater than 1 g/dL over 2 weeks can increase these risks. Avoid use in patients with a history of myocardial infarction, cerebrovascular event, or acute coronary syndrome within the 3 months prior to starting VAFSEO. Targeting a Hb level of greater than 11g/dL is expected to further increase the risk of death and arterial and venous thrombotic events, as occurs with ESAs, which also increase erythropoietin levels. No specific Hb target level, dose of VAFSEO, or dosing strategy has been identified to avoid these risks. Use the lowest effective dose and adhere to dosing and Hb monitoring recommendations to avoid excessive erythropoiesis. Advise patients to seek immediate medical attention if they develop signs or symptoms of myocardial infarction, stroke, venous thromboembolism, or thrombosis of vascular access. Evaluate and manage promptly if these occur. Hepatotoxicity Hepatocellular injury attributed to VAFSEO was reported in less than 1% of patients, including one severe case with jaundice. All events were asymptomatic and resolved after discontinuation of VAFSEO. The time to onset was generally within the first 3 months of treatment. Elevated serum ALT, AST, and bilirubin levels were observed in 1.8%, 1.8%, and 0.3% of CKD patients treated with VAFSEO, respectively. Measure ALT, AST, and bilirubin before treatment and monthly for the first 6 months, then as clinically indicated. Discontinue VAFSEO if ALT or AST is persistently elevated or accompanied by elevated bilirubin. Not recommended in patients with cirrhosis or active, acute liver disease. Hypertension Worsening of hypertension was reported in 14% (9.4 per 100 person-years [PY]) of patients receiving VAFSEO and 17% (11.8 per 100 PY) of patients receiving darbepoetin alfa. Serious worsening of hypertension was reported in 2.7% (1.7 per 100 PY) of patients receiving VAFSEO and 3% (1.8 per 100 PY) of patients receiving darbepoetin alfa. Cases of hypertensive crisis including hypertensive encephalopathy and seizures have also been reported in patients receiving VAFSEO. Monitor blood pressure. Adjust anti-hypertensive therapy as needed. Seizures Seizures occurred in 1.6% (1.0 per 100 PY) of patients who received VAFSEO and 1.6% (1.0 per 100 PY) of patients who received darbepoetin alfa. Following initiation of VAFSEO, monitor patients closely for premonitory neurologic symptoms. Monitor for new-onset seizures, premonitory symptoms, or change in seizure frequency. IMPORTANT SAFETY INFORMATION about VAFSEO (vadadustat) tablets (continued) 23

IMPORTANT SAFETY INFORMATION about VAFSEO (vadadustat) tablets (continued) 24 Gastrointestinal Erosion Gastric or esophageal erosions occurred in 6.4% (4.0 per 100 PY) of patients receiving VAFSEO and 5.3% (3.3 per 100 PY) of darbepoetin alfa-treated patients. Serious gastrointestinal (GI) erosions, including GI bleeding and the need for red blood cell transfusions were reported in 3.4% (2.1 per 100 PY) and 3.3% (2.0 per 100 PY) of those receiving VAFSEO and darbepoetin alfa, respectively. Consider the risk of GI erosion in high-risk patients, including those with a history of GI erosion, peptic ulcer disease, and tobacco or alcohol use. Advise patients of the signs and symptoms of erosions and GI bleeding and urge them to seek prompt medical care if present. Serious Adverse Reactions in Patients with Anemia Due to Chronic Kidney Disease and Not on Dialysis The safety of VAFSEO has not been established for the treatment of anemia due to CKD in adults not on dialysis and its use is not recommended in this setting. In large clinical trials in adults with anemia of CKD who were not on dialysis, an increased risk of mortality, stroke, myocardial infarction, serious acute kidney injury, serious hepatic injury, and serious GI erosions was observed in patients treated with VAFSEO compared to darbepoetin alfa. Malignancy VAFSEO has not been studied and is not recommended in patients with active malignancies. Malignancies were observed in 2.2% (1.3 per 100 PY) of patients treated with VAFSEO and 3.0% (1.8 per 100 PY) of patients treated with darbepoetin alfa. No evidence of increased carcinogenicity was observed in animal studies. ADVERSE REACTIONS The most common adverse reactions (occurring at ≥ 10%) were hypertension and diarrhea. DRUG INTERACTIONS Iron supplements and iron-containing phosphate binders: Administer VAFSEO at least 1 hour before products containing iron. Non-iron-containing phosphate binders: Administer VAFSEO at least 1 hour before or 2 hours after non-iron-containing phosphate binders. BCRP substrates: Monitor for signs of substrate adverse reactions and consider dose reduction. Statins: Monitor for statin-related adverse reactions. Limit the daily dose of simvastatin (20 mg) and rosuvastatin (5 mg). USE IN SPECIFIC POPULATIONS Pregnancy: May cause fetal harm. Lactation: Breastfeeding not recommended until two days after the final dose. Hepatic Impairment: Not recommended for use in patients with cirrhosis or active, acute liver disease. Please note that this information is not comprehensive. Please click here for the Full Prescribing Information, including BOXED WARNING and Medication Guide.

SOURCES 25 1 USRDS (https://usrds-adr.niddk.nih.gov/2022/end-stage-renal-disease/1-incidence-prevalence-patient-characteristics-and-treatment-modalities); DOPPS (https://www.dopps.org/DPM/DPMSlideBrowser.aspx); Based on internal estimates and industry reports estimating ESA pricing 2 https://www.asn-online.org/education/kidneyweek/2025/program-abstract.aspx?controlId=4352988 3 Phase 2 studies and Akebia preclinical studies (data on file with Akebia) 4 Preclinical studies and models (data on file with Akebia) 5 Nephcure/ The PARASOL Project: https://nephcure.org/the-parasol-project/ 6 Nephcure Kidney International: https://nephcure.org/wp-content/uploads/2021/02/nc.factSheet.FSGS_210106.pdf 7 Wedbush, Industry Note: “The IgAN Era Continues, Will FSGS Frenzy Follow?,” May 7, 2025 8 LifeSci Capital Alpha Series, Eledon Pharmaceuticals, Initiating Coverage, March 18, 2021 9 National Organization for Rare Diseases, Focal Segmental Glomerulosclerosis, November 21, 2018: https://rarediseases.org/rare-diseases/focal-segmental-glomerulosclerosis/ 10 C3 glomerulopathy - understanding a rare complement-driven renal disease. Nat Rev Nephrol. 2019 Mar;15(3):129-143. doi: 10.1038/s41581-018-0107-2. PMID: 30692664; PMCID: PMC6876298. https://pmc.ncbi.nlm.nih.gov/articles/PMC6876298/ 11 Evercore ISI, Travere Therapeutics, Inc. October 31, 2024 12 Jefferies Equity Research, Novartis AG, October 26, 2025 13 Systematic Review and Metaanalysis of Worldwide Incidence and Prevalence of Antineutrophil Cytoplasmic Antibody (ANCA) Associated Vasculitis. J Clin Med. 2022 May 4;11(9):2573. doi: 10.3390/jcm11092573. PMID: 35566698; PMCID: PMC9106044 14 Oppenheimer, Equity Research, Climb Bio, Inc. June 6, 2025 15 Cheruku SR, Raphael J, Neyra JA, Fox AA. Acute Kidney Injury after Cardiac Surgery: Prediction, Prevention, and Management. Anesthesiology. 2023 Dec 1;139(6):880-898. doi: 10.1097/ALN.0000000000004734. PMID: 37812758; PMCID: PMC10841304
